In the pharma and biotech industries, protein kinases are mainly produced using baculovirus with only a small fraction amenable to production using E.coli. The reason for this is twofold: i) E.coli. production often does not result in high levels of soluble protein for this conformationally flexible class of proteins ii) the majority of protein kinases are inactive if produced in E.coli since they lack the post translational modification (phosphorylation) required for activity. Due to cost versus yield, mammalian protein expression systems are hardly ever used for commercial production of kinases.
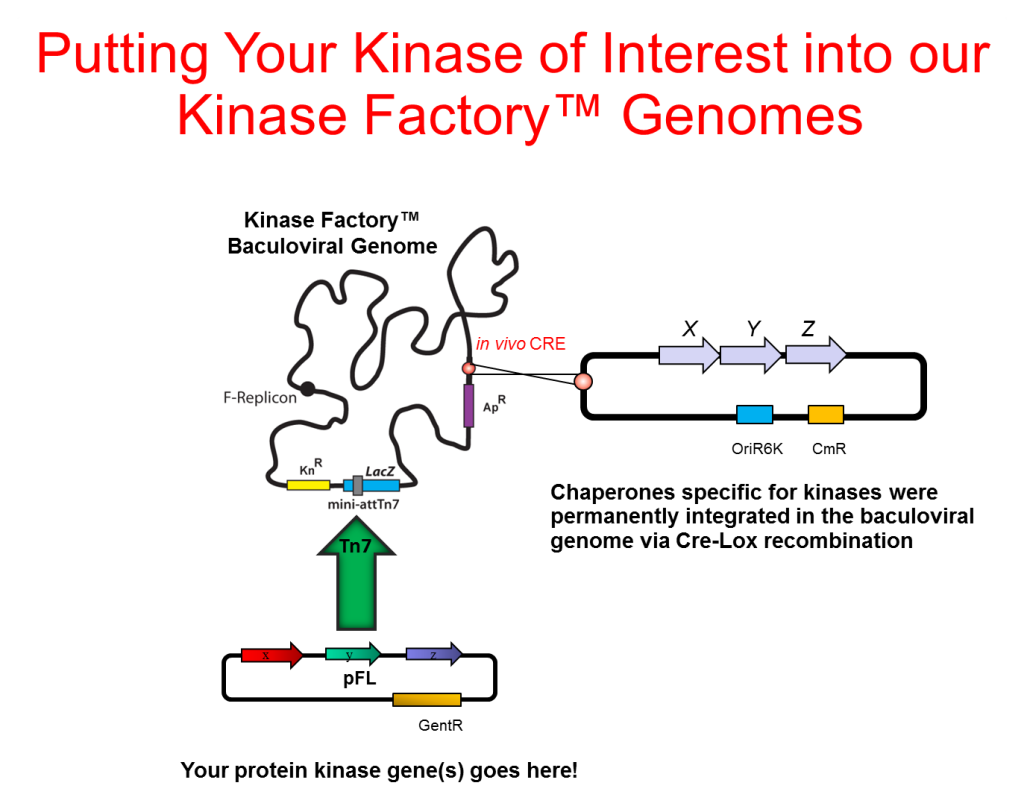
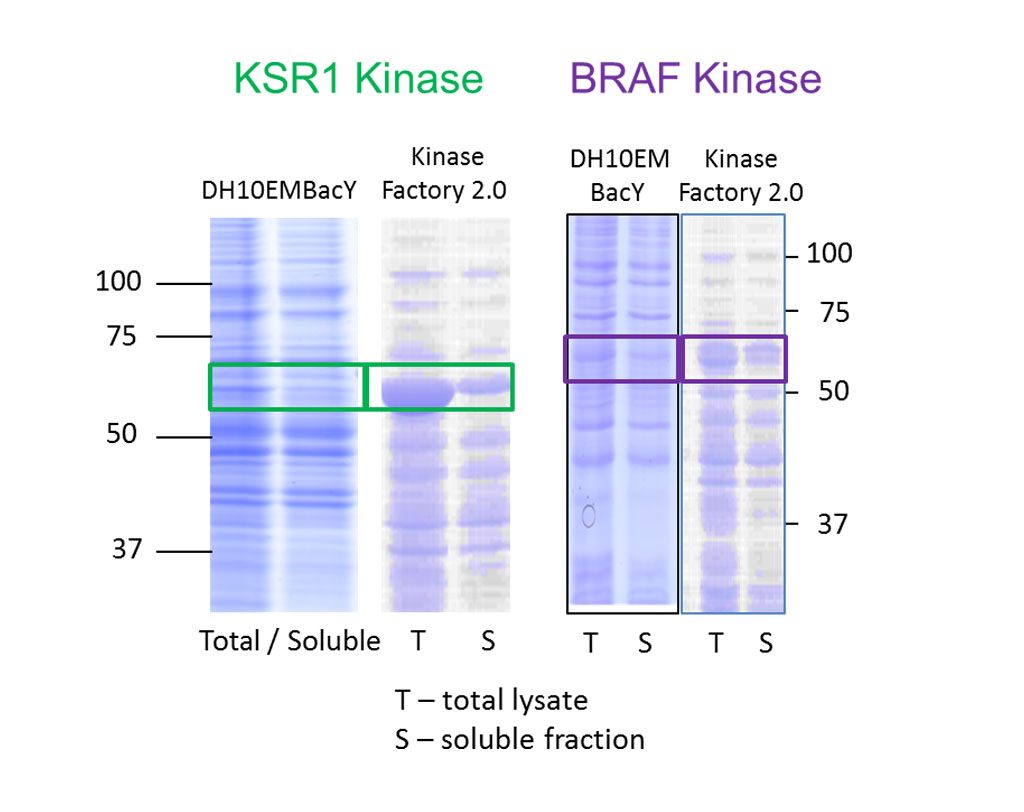
We have developed baculoviral genomes with integrated copies of entire chaperone machineries comprising up to 7 co-expressed chaperone proteins built around a Hsp90 core. The components were selected from the literature to boost solubility and yield of a broad variety of human protein kinases.
Use MultiBac™ protocols (click here) to assemble Kinase Factory™ Viruses
Click here for DNA vector sequences
Click here to download Cre-ACEMBLER User Manual 3.0
Click here to download Cre-ACEMBLER Short Description