Virus-like particles (VLPs) are multiprotein structures that mimic the organization and conformation of authentic native viruses but lack the viral genome, making them non-infectious and safe to handle. VLPs are becoming an increasingly widespread tool for the development of vaccines, and are finding other important applications in biotechnology including serving as a display system for integral membrane proteins for antibody screening, production of immunogens, and for ligand binding assays. The expression of viral structural proteins, such as matrix proteins, can result in the self-assembly of VLPs.
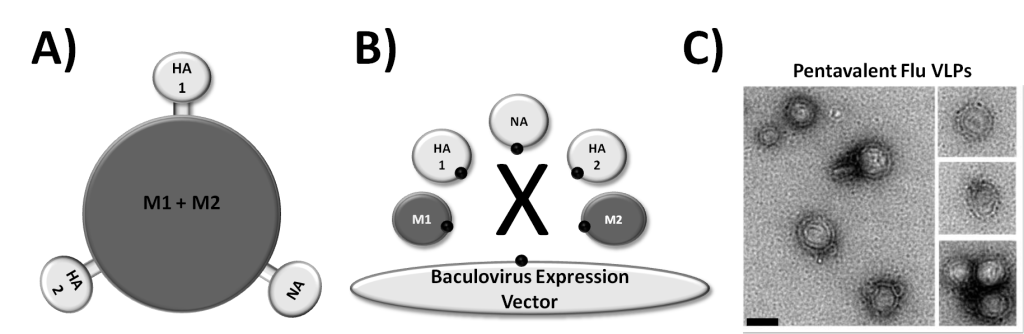
Figure 1. Assembly of VLPs (left and middle). Electron Micrographs of Pentavalent Flu VLP Vaccine Candidate Created Using VLP Factory™ (right).
VLP Factory™ is a baculovirus expression system which constitutively secretes promiscuous virus matrix proteins, with built in functionality to allow you to introduce multiple additional expression cassettes for production of multiprotein VLPs displaying your surface protein, or integral membrane protein, of choice.
VLP Factory™ transfer vectors are fully synthetic, comprising only functional DNA, and as a result are small and easy to handle. The VLP Factory™ baculovirus genome (DH10VLPFactory) is under continuous development to remove harmful/non-essential genes (e.g. v-cath and chiA) and DNA recombination hotspots.
Applications for multiprotein VLPs include their use directly as as multiepitope vaccines, for ligand binding assays, as templates for small molecule drug development, as templates for therapeutic antibody development via phage or yeast display, for screening for and characterizing monoclonal antibodies, as carriers for membrane proteins e.g. GPCRs or ion channels, as adjuvants or for prime-boost regimens, or as scaffolds for synthetic metabolic pathways (substrate channeling).
Click here to download VLP Factory™ Manual v1.2
Click here for DNA vector sequences
Click here to download Cre-ACEMBLER User Manual 3.0
Click here to download Cre-ACEMBLER Short Description